 The following equation gives the quantitative relationship between the original number of nuclei present at time zero (N_0\) and the number (\(N\)) at a later time \(t\). The term is commonly used in nuclear physics to describe how quickly unstable atoms undergo radioactive decay or how long stable atoms survive. 
It is also applicable to the decay of excited states in atoms and nuclei. Half-life (symbol t) is the time required for a quantity (of substance) to reduce to half of its initial value. The concept of half-life is applicable to other subatomic particles, as will be discussed in Particle Physics. Nuclides with the shortest half-lives are those for which the nuclear forces are least attractive, an indication of the extent to which the nuclear force can depend on the particular combination of neutrons and protons. 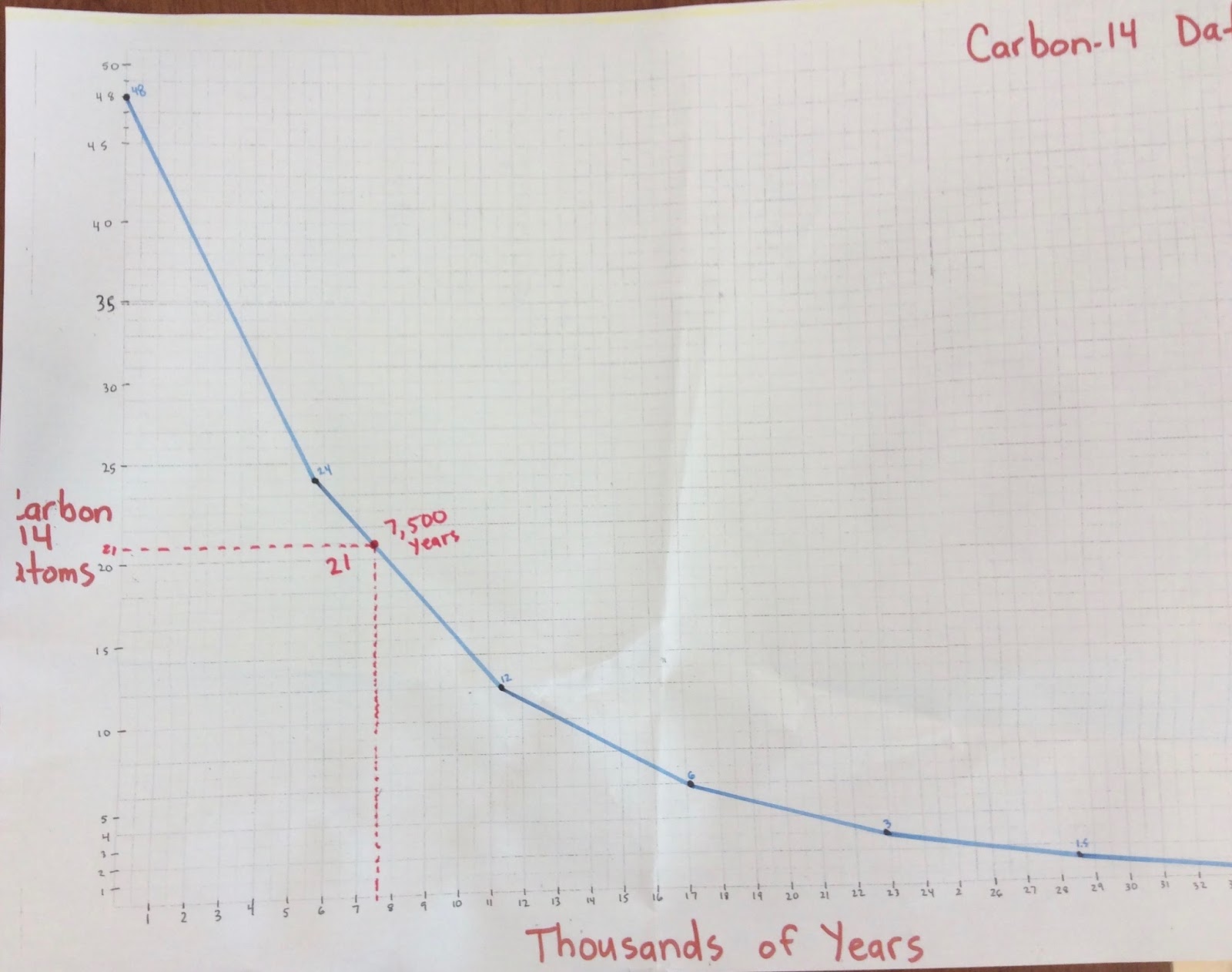
\)y for the least unstable, or about 46 orders of magnitude. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
